Best Peptides for Fat Loss: 2026 Comparison Guide

Built and maintained by Garret Grant - Founder & Lead Researcher, B.S. Engineering, UCLA.
Last updated: May 2, 2026
Human-researched and AI-assisted with full editorial review. I verify sources, rankings, and final judgments personally. See methodology.
Share this page
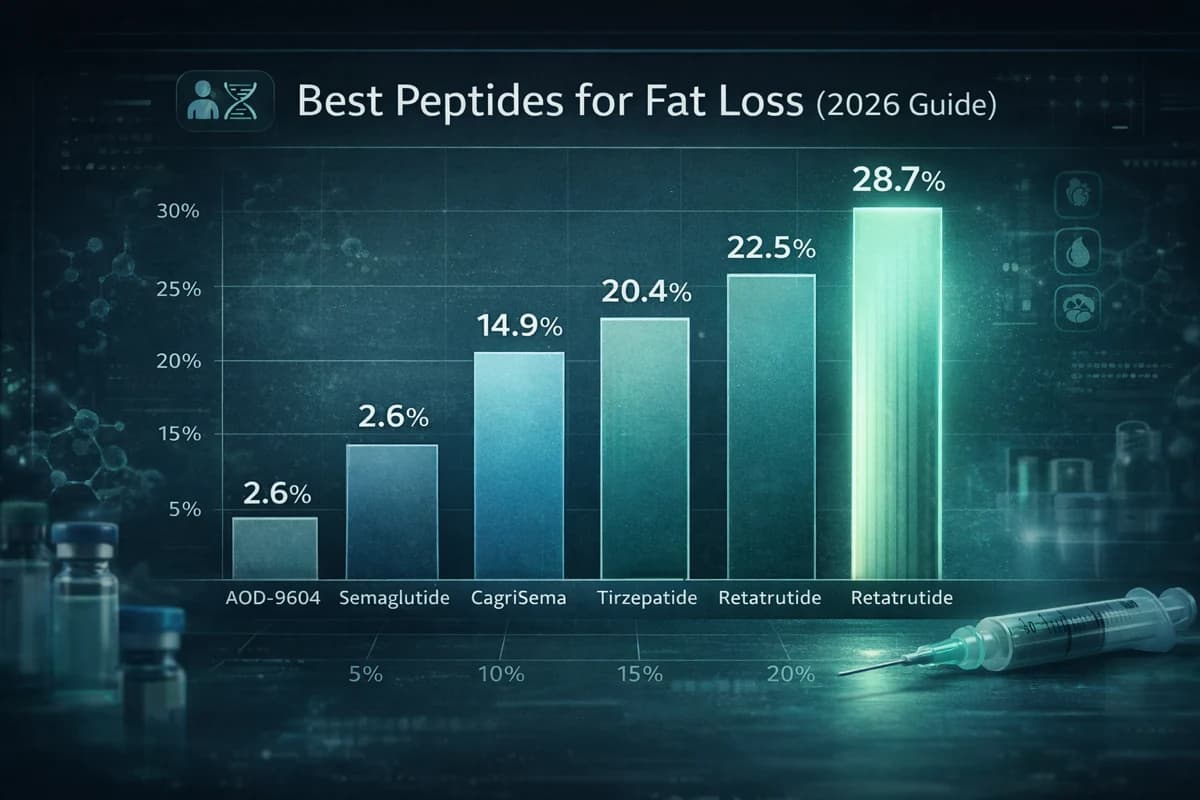
Retatrutide is the clear top performer for fat loss in 2026, with GLP-1 receptor agonists and their multi-receptor successors dominating the category. Semaglutide produces roughly 14.9% to 15.2% mean weight loss, tirzepatide delivers 16% to 22.5%, retatrutide achieved 24.2% to 28.7% in Phase 2/3 data, and CagriSema showed 20.4% in Phase 3. AOD-9604 and MOTS-c offer different, less-proven mechanisms. This guide compares all seven compounds on efficacy, safety, dosing, cost, and supplier availability.

Fat-loss peptides have moved far beyond marketing hype. The STEP, SURMOUNT, TRIUMPH, and REDEFINE programs now provide specific, peer-reviewed weight-loss percentages that allow direct comparison. This guide evaluates seven peptides across clinical efficacy, side effects, sustainability concerns including muscle loss and rebound weight gain, reconstitution math, current pricing from COA-verified suppliers, and access pathways including clinical trial enrollment.
Whether evaluating research-grade options through PepPal's verified suppliers or considering clinical trial participation, every claim in this guide is anchored to published trial data or clearly labeled as community-derived. For compound-specific dosing and titration details, see the protocol library on peptidedosingprotocols.com.
Disclaimer: This article is for educational and informational purposes only. It is not medical advice. Regulatory status varies by compound. Semaglutide and tirzepatide are FDA-approved, while retatrutide and CagriSema remain investigational, and AOD-9604 plus MOTS-c are research-use compounds only. Consult a healthcare provider before considering any compound.