
Peptide Partners
Our #1 supplier
Same-day FedEx 2-Day Air, 59 Finnrick tests, multi-lab coverage, and the best per-mg value make Peptide Partners the recommended source for most buyers.
TL;DR top pick
Peptide Partners is PepPal's current #1 pick for most buyers because it has the deepest Finnrick sample set in this guide, multi-lab documentation, credit card acceptance, and strong per-mg value for repeat orders.
Finding the best grey market peptide supplier in 2026 requires more than reading a product page or copying a discount code. This guide ranks 10 suppliers using Finnrick Analytics testing data, Janoshik COA verification, and community trust signals — making it the most comprehensive grey market peptide vendor comparison available. The supplier landscape shifted dramatically this year. Peptide Sciences, previously the largest grey market vendor in the U.S., shut down on March 6, 2026 without warning. Amino Asylum was raided by the FDA. At least seven vendors closed in the past twelve months. The market is smaller, the remaining vendors are under more scrutiny, and verification discipline matters more than ever.
To build this comparison, I reviewed active vendor pages, pulled Finnrick vendor and per-compound ranking data dated through April 10, 2026, checked available Janoshik and batch COA records, and compared those findings against community discussion patterns and buyer workflow issues showing up in PepPal's calculator usage.
I reviewed 10 supplier profiles for this guide, cross-referencing Finnrick Analytics test data covering 6,195 samples across 185 vendors, Janoshik verification reports, batch-specific COA documentation, and community trust patterns from Reddit, Trustpilot, and long-running Telegram groups including STG and Peptide Research Group. I also drew on usage patterns from over 10,000 PepPal calculator sessions to identify where buyers most commonly make sourcing and reconstitution mistakes. Every recommendation below maps back to concrete buyer actions so you can validate claims yourself before spending money.
This guide includes non-affiliate entries and benchmark context so the rankings are not skewed by commission relationships. Use it as a starting framework and run your own verification on every order. No supplier ranking replaces batch-level due diligence.
For compound-level dosing context before buying, review the Retatrutide research protocol and the supplier directory for side-by-side reviews. New to peptides entirely? Start with the complete peptide list to understand what is available and what each compound does.
Research-use-only notice
This page is educational and does not provide medical advice. Grey market peptides are not FDA-approved for human consumption. Always validate legal, quality, and safety requirements before purchasing research materials.
Editorial independence
I apply the same published scoring rubric to every supplier in this guide, checking Finnrick data, COA documentation, payment and fulfillment signals, and repeat community patterns before assigning a rank. Some links are affiliate links and are marked, but affiliate status does not change score weighting.
These are PepPal's recommended supplier options, both USA-based and offering the PEPPAL discount code at checkout when eligible. For a side-by-side breakdown of our two recommended suppliers, review the supplier directory.

Peptide Partners
Our #1 supplier
Same-day FedEx 2-Day Air, 59 Finnrick tests, multi-lab coverage, and the best per-mg value make Peptide Partners the recommended source for most buyers.

Pivot Labs
Need another option?
Fast domestic fulfillment, practical product coverage, and straightforward ordering make Pivot Labs a useful alternate supplier.
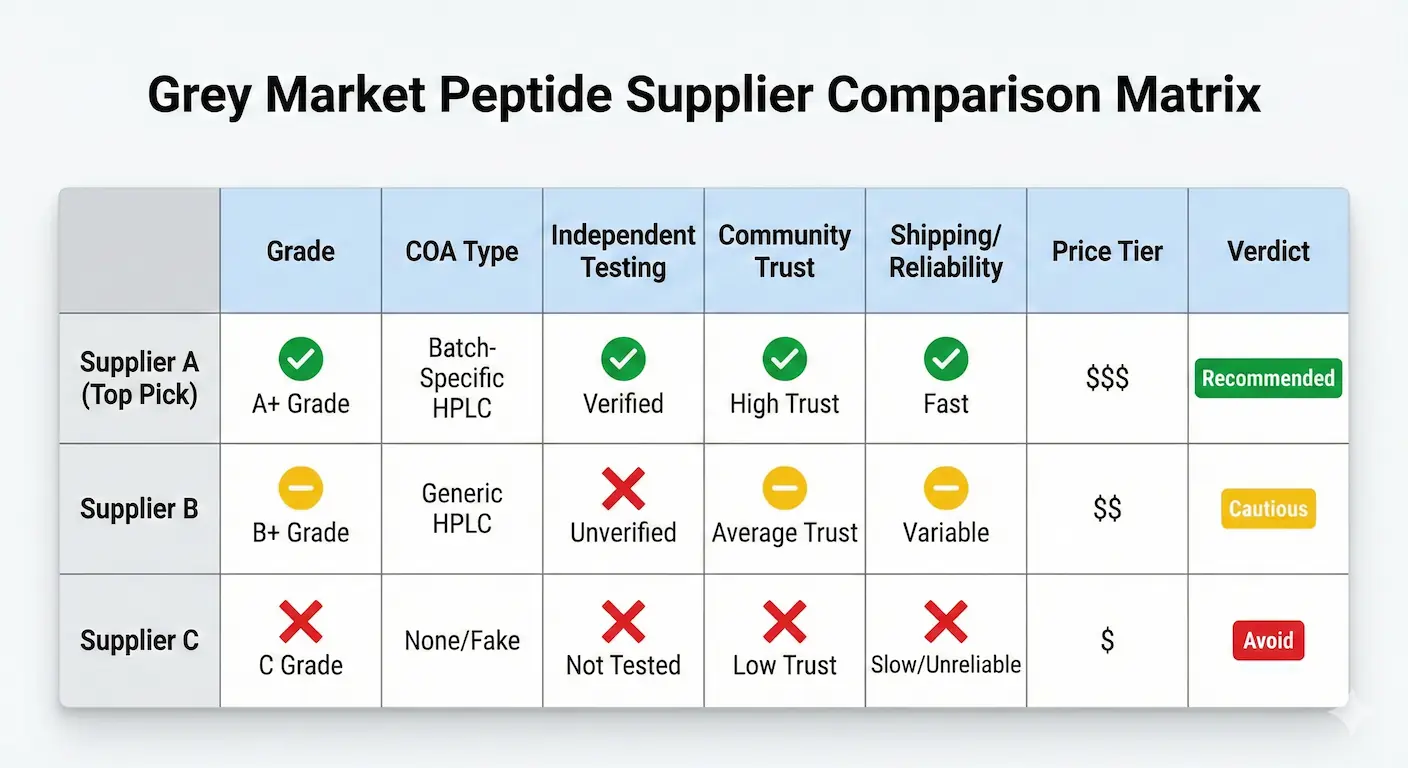
Rankings below are structured for practical buying decisions, not promotional copy. Every profile follows the same framework: verification signal strength, COA transparency, trust indicators, fulfillment reliability, and value at current pricing levels. Every supplier is scored with the same rubric.
For each ranked profile, I reviewed the supplier's live product and policy pages, checked whether COA access was batch-specific or generic, compared Finnrick and Janoshik visibility where available, and separated directly documented signals from reputation patterns inferred through repeat community discussion.
Rank #1
Grade A
COA quality: Batch-linked purity reports with additional endotoxin, heavy-metals, and sterility documentation. Janoshik-verified COAs published for every product. Multi-lab coverage provides redundant quality signals.
Community reputation: 59 Finnrick-tested samples across 7 products: the highest independent sample depth of any vendor in this guide. Frequently cited in Reddit r/Peptides supplier threads, STG Telegram, and Peptide Research Group discussions.
Fulfillment: Same-day FedEx 2-Day Air shipping on most orders. USA-based warehouse. Credit card and debit card accepted (payment processor compliance verified). Consistent support response times and repeat-order reliability. Operational since 2021.
Pricing tier: $$: Value-forward when factoring larger vial formats and combo-stack options. Per-mg cost is competitive with or lower than most Finnrick A-rated alternatives. Use code PEPPAL for available discount.
Best for: Researchers prioritizing maximum verification depth, multi-lab quality signals, and stable reorder confidence. Best bulk and full-cycle value.
PepPal verdict: #1 overall. Highest Finnrick sample depth, Janoshik-verified COAs, credit card acceptance, and competitive per-mg pricing make Peptide Partners the default recommendation for most buyers.
Rank #2
Grade COA review
COA quality: Batch COA review and supplier-provided documentation should be checked against the exact product and lot before purchasing. Best used with the same lot-matching workflow applied to Finnrick-listed vendors.
Community reputation: Active PepPal secondary supplier with domestic shipping, responsive support positioning, and PEPPAL code support.
Fulfillment: Fast domestic shipping emphasis. Confirm current checkout, payment, and replacement terms on the supplier site.
Pricing tier: $$: Mid-tier pricing. Use code PEPPAL when eligible.
Best for: Researchers who want a practical Peptide Sciences replacement with domestic shipping, broad research peptide access, and PepPal discount-code access.
PepPal verdict: #2 overall. Active secondary option for buyers who want practical catalog coverage and straightforward ordering after Peptide Partners.
Rank #3
Grade A-
COA quality: Moderate documentation depth with improving transparency. Public test visibility is less broad than the strongest A-rated vendors. Buyers should request batch-specific COAs directly and verify lot number matching.
Community reputation: Legacy reviewed supplier with broad research compound coverage and useful comparison value for buyers checking multiple sources after Peptide Partners and Pivot Labs.
Fulfillment: USA domestic shipping, 3–6 business days. Payment methods include credit card. Generally dependable for standard domestic workflows, though fulfillment speed may vary by period.
Pricing tier: $–$$: Budget-to-mid pricing that can be attractive when buyers accept the extra due diligence overhead required with less documented vendors.
Best for: Researchers comparing broader catalog access after checking Peptide Partners and Pivot Labs first. Useful as a comparison source when a specific compound is not available elsewhere.
PepPal verdict: #3 overall. Strong catalog-depth comparison option, but still requires batch-level verification discipline. Confirm every COA matches your vial lot before use.
Rank #4
Grade A-
COA quality: Publicly available COAs with batch-specific documentation. Third-party testing confirmed across core products. Testing coverage is narrower than the top-ranked suppliers but consistent where documented.
Community reputation: Regularly appears in shortlists for GLP-1-focused buying on Reddit and comparison forums. Repeat mentions for quality-to-price value. Note: Paradigm Peptides' founders Matthew Kawa and Jennifer Stechkober pleaded guilty to federal charges on December 10, 2025. Federal investigators determined their SARM products contained testosterone, a controlled substance, and brought additional charges for selling unapproved drugs without FDA authorization.[6] This prosecution involved their SARM product line, not their peptide catalog, but buyers should be aware of the legal history and monitor for any operational changes.
Fulfillment: USA domestic shipping, 2–5 business days. Credit card accepted. Solid fulfillment consistency based on community-reported buying workflows.
Pricing tier: $–$$: Often positioned as a value-forward option, particularly for GLP-1 peptides. Per-vial cost is frequently lower than the co-leaders at comparable quality tiers.
Best for: Researchers who want strong quality indicators while keeping per-vial cost tightly managed. Good option when budget is a primary factor.
PepPal verdict: High-value contender and a realistic alternative when cost sensitivity is higher. Monitor legal developments given the 2025 SARM prosecution.
Rank #5
Grade B
COA quality: Supplier-level transparency is discussed in community channels, but COA coverage varies by product line. Buyers should verify each batch record directly and confirm whether independent third-party testing backs the documentation.
Community reputation: Known in Reddit and forum comparisons for fast fulfillment speed and responsive ordering experience. Sentiment is mixed but generally solid. Fewer independent Finnrick data points are available compared to higher-ranked vendors.
Fulfillment: USA domestic shipping, 1–4 business days, typically among the fastest in this guide. Strong attention to support communication and tracking.
Pricing tier: $$: Not the lowest price, but often justified by operational consistency and support reliability.
Best for: Researchers who prioritize fulfillment speed and support responsiveness over chasing the lowest list pricing.
PepPal verdict: Reliable operational profile. Best used after confirming lot-specific documentation quality for your specific compound. Included for comparison completeness.
Rank #6
Grade B
COA quality: Third-party COAs available on their website, though the testing laboratory is not publicly named. Finnrick has tested 22 samples across 4 products; results are mixed. BPC-157 earned a Finnrick B rating (Good) across 6 samples. However, CJC-1295 received a Finnrick D rating (Poor) across 8 samples, and Ipamorelin also received a D rating. This product-level variance means buyers must check Finnrick data for their specific compound, not assume the vendor grade applies uniformly.
Community reputation: Polarized reviews across platforms: 4.5/5 on Trustpilot (150+ reviews) but lower ratings on Sitejabber. Popular in the r/Peptides community for nootropic and recovery compounds. Founded by a healthcare professional in Gulf Breeze, Florida. Offers injectable vials, oral capsules, and nasal spray formats.
Fulfillment: USA domestic shipping. Free shipping threshold is $350, higher than most competitors. Accepts credit card, cryptocurrency (10% discount), CashApp (5% discount), and bank transfer.
Pricing tier: $$–$$$: Mid-to-premium pricing. BPC-157 10mg equivalent runs approximately $94 before discounts. Payment method discounts (crypto, CashApp) can offset the premium.
Best for: Researchers interested in alternative formats (oral capsules, nasal sprays) or nootropic-focused compounds like Semax, Selank, and Dihexa that may be harder to find from other vendors.
PepPal verdict: Format diversity and nootropic selection are genuine differentiators. But the mixed Finnrick results across products mean you must verify quality data for each specific compound. Do not assume a passing BPC-157 grade extends to CJC-1295 or Ipamorelin.
Rank #7
Grade B-
COA quality: COAs are available on request rather than prominently displayed on product pages. Testing documentation is less transparent than vendors that publish COAs directly. Buyers must contact support to obtain documentation before purchasing. No Finnrick listing at time of writing.
Community reputation: Budget-friendly and popular for bulk orders in Reddit discussions. Quality is generally reported as adequate in community threads, but documentation transparency lags behind top-tier vendors. Less discussion volume than higher-ranked suppliers.
Fulfillment: USA domestic shipping. Standard delivery timelines. Payment methods include credit card.
Pricing tier: $: Positioned as a budget-friendly option for researchers ordering in bulk. Per-vial pricing is typically lower than Finnrick A-rated vendors.
Best for: Researchers who are experienced with independent verification, comfortable requesting documentation manually, and prioritizing cost above all other factors.
PepPal verdict: Lowest-cost option in this guide, but the reduced documentation visibility means extra verification work falls on the buyer. Not recommended as a sole source. Better as a secondary vendor alongside a primary A-rated supplier.
Rank #8
Grade B-
COA quality: Claims third-party testing but provides limited public documentation about which labs perform the tests and how frequently. Not currently listed on Finnrick. COAs are available for some products but coverage varies.
Community reputation: Las Vegas-based vendor. 4.8/5 Trustpilot rating. Known for catalog breadth: carries approximately 50 compounds spanning metabolic peptides, tissue repair compounds, and growth hormone secretagogues. Community sentiment is mixed, with some users flagging inconsistencies in COA documentation across products.
Fulfillment: USA domestic shipping, 3–5 business days typical. Credit card accepted. Standard support responsiveness.
Pricing tier: $–$$: Mid-range pricing. Approximately $35–75 per vial depending on compound. Below premium vendors like the former Peptide Sciences but above budget-tier suppliers.
Best for: Researchers who need a wide catalog selection and are willing to do their own third-party verification for each batch. Useful when sourcing obscure or specialty compounds.
PepPal verdict: Catalog breadth is a genuine advantage for hard-to-find compounds. But the absence of Finnrick listing and inconsistent COA visibility mean buyers carry more verification burden.
Rank #9
Grade A (compound-specific)
COA quality: Varies by vendor. Buyers should request batch-specific documentation directly and verify it against Finnrick data for their target compound.
Community reputation: These vendors appear in Finnrick's per-compound rankings but have smaller community discussion footprints than the top-8 profiled suppliers.
Fulfillment: Varies by vendor. Confirm shipping methods, payment options, and return policies directly before ordering.
Pricing tier: $$–$$$: Pricing varies by compound and seller. These are worth checking when a specific compound has standout Finnrick performance, but price should remain secondary to verification.
Best for: Researchers targeting specific compounds where these vendors hold top Finnrick ratings. Always verify current Finnrick data before ordering because ratings are compound-specific and can change between batches.
PepPal verdict: Worth investigating for compound-specific sourcing, but they require more buyer-side verification than the fully profiled vendors above.
Rank #10
Grade Unrated
COA quality: Variable. Some marketplace sellers provide Janoshik-verified COAs; many do not. The burden of requesting and verifying documentation is entirely on the buyer, so pre-purchase COA verification is essential.
Community reputation: Active discussion exists in Telegram groups such as STG and Peptide Research Group, plus Reddit threads and community testing groups. Quality variance between marketplace sellers is high, so one passing order does not predict the next.
Fulfillment: International shipping typically takes 1-3 weeks from China. Most payment methods lack credit card chargeback protection, and cold-chain integrity during transit is uncertain.
Pricing tier: $: Significantly lower than US-based suppliers. Per-mg cost can be 3-5x lower on compounds like retatrutide and tirzepatide, but the verification burden rises sharply with the lower price.
Best for: Experienced researchers with independent testing workflows who prioritize cost above all other factors and are willing to manage the additional verification, shipping, and quality-control burdens.
PepPal verdict: Not recommended for most buyers. The cost savings are real, but the key risks are longer shipping times, limited recourse for failed orders, customs exposure, uncertain cold-chain management, and higher contamination risk without verified clean-room standards. If you are researching a specific compound first, cross-reference PepPal's complete peptide directory before chasing a marketplace seller.
Rank Benchmark
Grade C
COA quality: Generic or recycled COAs are common, with limited third-party validation traceability.
Community reputation: Trust signals are fragmented across forums, often with inconsistent identity and domain history.
Fulfillment: Fulfillment reliability is highly variable, especially for returns, replacements, and policy enforcement.
Pricing tier: Low pricing can be attractive but frequently coincides with weaker quality controls and higher uncertainty.
Best for: Only for advanced buyers running strict independent verification workflows every order cycle.
PepPal verdict: This benchmark highlights why objective verification matters more than price-first buying.
For readers focused on specific compounds, see the Retatrutide access guide and the supplier directory for a broader market scan.
I compiled this matrix from the reviewed supplier profiles, current vendor pages, Finnrick context, and the source set cited below so buyers can compare the same decision points across 10+ vendors and marketplace models in one place. The overall grade is directional, while the Finnrick context column shows where compound-specific strength is strongest or where blanket vendor assumptions break down.
If the last columns are cut off on your screen, scroll horizontally to view the full matrix.
| Supplier | Overallgrade | Finnrickcontext | COAtype | Third-partytesting | Paymentmethods | Trustsignal | Shippingspeed | Est. pricerange | PepPalverdict |
|---|---|---|---|---|---|---|---|---|---|
| Peptide Partners | A (59 samples) | Deepest sample set; strongest published BPC-157 and Ipamorelin context. | Batch-linked + multi-lab | Janoshik + Finnrick verified | Credit card, debit | High activity | 2–4 days(same-day ship) | $$ | #1 (best bulk value) |
| Pivot Labs | COA review | Active PepPal secondary pick; verify product and lot documentation before checkout. | Batch COA review | Supplier COA review | Credit card | Active secondary | 2–5 days | $$ | #2 (practical catalog) |
| Orbitrex Peptides | A- | Legacy comparison option with broader catalog context in PepPal coverage. | Batch-specific | Partial Finnrick visibility | Credit card | Moderate | 3–6 days | $–$$ | #3 (catalog comparison) |
| Paradigm Peptides | A- | Top current retatrutide context; strong GLP-1 value positioning. | Batch-specific | Third-party confirmed | Credit card | Moderate-high | 2–5 days | $–$$ | Best value contender |
| Peptide Tech | B | Varies by product; verify each compound individually. | Mixed by product | Varies | Credit card | Mixed | 1–4 days | $$ | Speed-focused profile |
| Limitless Life Nootropics | B (mixed by product) | BPC-157 performs better than CJC-1295 and Ipamorelin. | Third-party available | 22 Finnrick samples (B–D range) | CC, crypto, CashApp | Polarized (4.5 TP) | 3–5 days | $$–$$$ | Format diversity leader |
| Core Peptides | B- | Not Finnrick-listed; no compound-specific edge published. | On request | Not Finnrick-listed | Credit card | Limited | 3–5 days | $ | Budget bulk option |
| Biotech Peptides | B- | Catalog breadth matters more than Finnrick depth here. | Varies by product | Not Finnrick-listed | Credit card | Moderate (4.8 TP) | 3–5 days | $–$$ | Catalog breadth leader |
| Polaris / Nuscience / Skye | A (compound-specific) | BPC-157, Tirzepatide, Retatrutide, and Semaglutide appear in top-per-compound snapshots. | Varies by vendor | Finnrick per-compound leaders | Varies | Lower discussion volume | Varies | $$–$$$ | Compound-targeted option |
| International marketplace model | Unrated | Marketplace-dependent; seller-by-seller verification required every order. | Variable | Buyer-managed | Often crypto, wire | Community-dependent | 1–3 weeks | $ | High-risk low-cost path |
| Category baseline | C | No reliable compound-specific edge; verification burden is highest. | Generic/none | Rare | Often crypto-only | Fragmented | Unpredictable | $ | High verification burden |
Grey market peptides are sold outside traditional pharmaceutical distribution channels and labeled for research use only. In practical terms, that means buyers do not receive the regulatory protections, quality controls, or patient safeguards that exist in standard prescription supply chains. The burden of verification shifts directly to the buyer, which is exactly why this page focuses on repeatable validation rather than brand promises.
It helps to separate three categories that often get blurred in search results. Grey market suppliers operate in a legal gray zone where products are sold as research chemicals, not FDA-approved, not DEA-scheduled controlled substances, but not explicitly illegal to purchase. The FDA has stated that "research use only" disclaimers on peptide products sold with diluent and syringes are "a ruse to avoid FDA scrutiny for selling misbranded and adulterated products in violation of the FD&C Act." This enforcement position means the legal gray zone is narrower than many buyers assume. [3] Black market operators involve deliberate counterfeiting, mislabeling, or fraudulent channels with no identity continuity. Compounding pharmacies, by contrast, work inside regulated frameworks such as 503A and 503B, require prescription-driven workflows, and are subject to inspection requirements that grey market suppliers do not follow.
The demand surge around GLP-1-focused peptides like retatrutide and tirzepatide made this distinction more important than ever in 2026. Following the February 2026 HHS announcement restoring approximately 14 peptides to Category 1 compounding status, buyers now have a regulated access path for many compounds that were previously grey-market-only. At the same time, the FDA's enforcement actions against unapproved GLP-1 products have increased pressure on remaining grey market vendors. Search interest in both "grey market" and "gray market" phrasing keeps growing. These spelling variants describe the same core issue: access can be easier and cheaper, but verification discipline must increase in direct proportion. If you skip that discipline, lower cost can quickly become higher risk.
A reliable workflow starts with documentation hygiene, independent data checks, and storage/reconstitution planning. Before any order, confirm whether you can match lot numbers, verify COA authenticity, and reproduce concentration math in advance. PepPal's reconstitution walkthrough and calculator are built to support that process after supplier verification is complete. If you are considering stacking multiple compounds, verification matters even more because you are introducing multiple supply chain variables. Bought your peptides? Next step is baseline labs, so read the blood work guide.
The grey market peptide industry entered 2026 in a state of rapid consolidation. Understanding what happened over the past six months is essential context for evaluating which suppliers remain and why the landscape looks different from 2025.
To track that shift, I reviewed vendor shutdown notices, FDA enforcement updates, legal coverage on peptide compounding and enforcement, Finnrick testing changes, and visible supplier availability changes across the vendors covered in this guide.
Peptide Sciences shut down on March 6, 2026. The largest grey market vendor in the United States, reportedly generating approximately $7.4 million in monthly sales as of December 2025, posted a three-sentence notice announcing a voluntary closure and went dark. No refunds were processed. No advance warning was given. Finnrick Analytics had previously flagged quality issues with several Peptide Sciences products, including a failing E rating on retatrutide across 37 tested samples and counterfeit detection in November 2025. The closure left thousands of buyers searching for alternatives and redirected substantial demand to the remaining vendor pool.
Amino Asylum was raided by the FDA in June 2025. Federal agents seized inventory and shut down operations. This was the first physical raid on a major peptide vendor, escalating enforcement from warning letters to direct action.
Paradigm Peptides' founders pleaded guilty to federal charges in December 2025 after an investigation revealed their SARM products contained testosterone, a controlled substance. This prosecution demonstrated that federal penalties in this space are severe and not limited to warning letters.
The FDA issued 50+ warning letters to GLP-1 compounding pharmacies in a single month (September 2025), and the ITC issued exclusion orders blocking tirzepatide imports, further tightening the supply environment.
In a September 2025 analysis, Wilson Sonsini, one of the largest law firms in the U.S., documented that the FDA warning letters specifically targeted claims that compounded products are "generic versions" or contain the "same active ingredient" as FDA-approved GLP-1 drugs, language the FDA considers false or misleading.[1]
In April 2026, the FDA removed 12 peptides from Category 2 effective April 22, but that action did not add them to the 503A bulks list or authorize compounding. The current regulatory posture is still a holding pattern pending July 2026 PCAC review. For the full breakdown, see the April 2026 Category 2 removal explainer. That shift still does not change the grey market's verification requirements.
The FDA's own safety page on unapproved GLP-1 drugs notes that compounded drugs "are not FDA approved" and warns that the agency "has received reports of adverse events related to compounded versions of semaglutide and tirzepatide," including products arriving without adequate refrigeration and fraudulent labels listing pharmacies that did not actually compound the products.[2]
The net effect: fewer suppliers, higher demand on remaining vendors, and increased regulatory pressure across the board. Every supplier ranked in this guide is still operating as of April 2026, but the landscape can shift quickly. Treat any ranking as a snapshot, not a permanent endorsement, and re-verify before every purchase cycle.
After the Peptide Sciences shutdown on March 6, 2026, thousands of researchers needed new sources. To identify the strongest alternatives, I compared Finnrick testing data with post-shutdown Reddit and Telegram discussion trends, supplier comparison threads, and the vendors buyers most often cross-shopped in PepPal's supplier research workflow after March 6, 2026.
For buyers who valued Peptide Sciences' catalog breadth: Pivot Labs is the strongest fit because it combines a practical research peptide catalog with domestic shipping, PEPPAL code support, and a straightforward checkout workflow. Biotech Peptides remains relevant for buyers chasing maximum category coverage, but Pivot Labs is the better active secondary replacement for most PepPal readers.
For buyers who valued Peptide Sciences' testing reputation: Peptide Partners now has the deepest Finnrick sample set in the market with 59 tested samples and an A rating across 7 products. This exceeds the testing depth Peptide Sciences had at its peak, and Finnrick flagged Peptide Sciences' retatrutide with a failing E rating across 37 samples before the shutdown.[5]
For buyers who valued Peptide Sciences' pricing: Peptide Partners is the strongest fit because its cost per mg is excellent once you factor in larger vial formats, combo options, and the depth of verification backing the catalog. Paradigm Peptides still works as a lower-cost alternative in some categories covered in PepPal's fat loss peptide guide, but the cheapest-looking replacement is not necessarily the best. The company's closure coincided with documented quality issues, which underscores why independent testing data matters more than brand familiarity.
For a full breakdown of each alternative, see the ranked supplier profiles below or the supplier directory for detailed reviews.
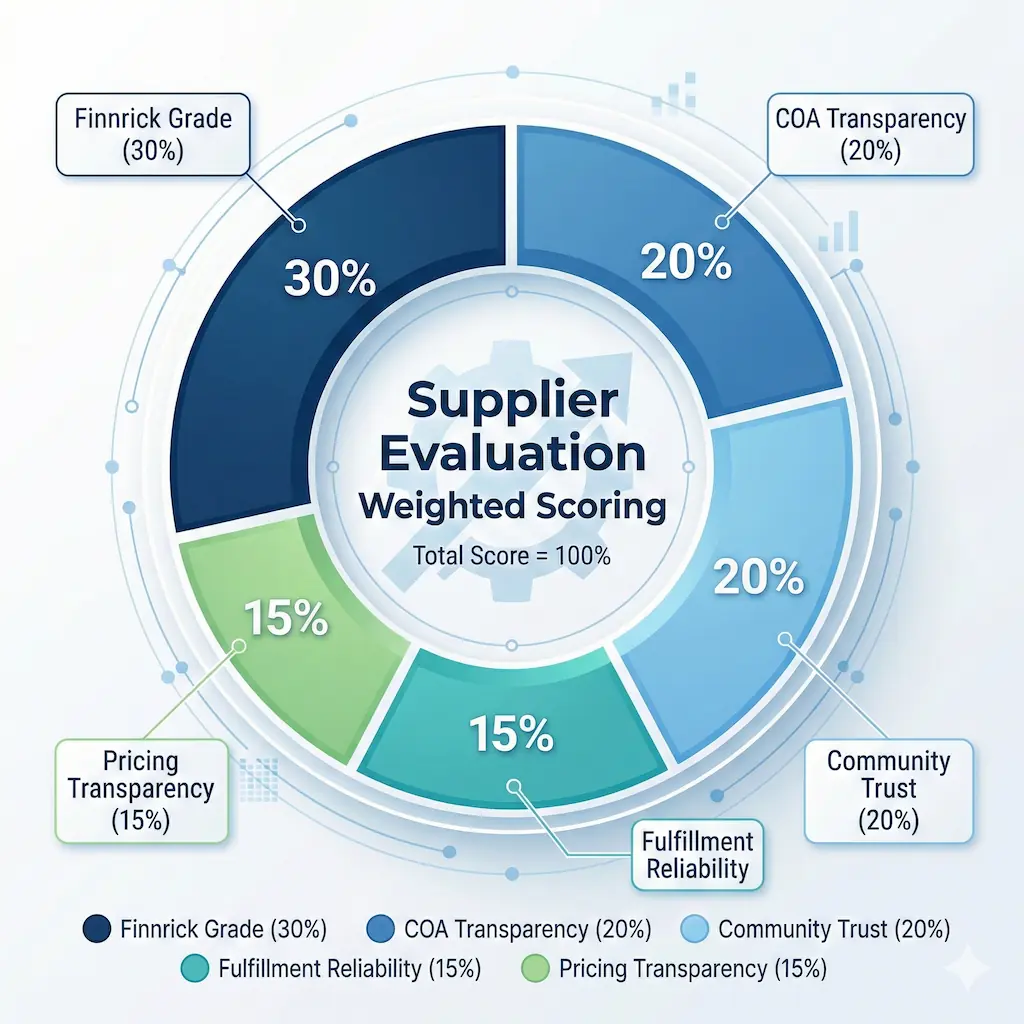
The strongest ranking guides are transparent about how a vendor earns position. I built this weighted rubric to reduce bias and make rankings repeatable across update cycles, and I refresh it as new testing data, supplier policy changes, or documentation gaps appear. I score each supplier against five criteria, then run an editorial review pass that checks whether each claim can be traced to documentation or stable community evidence before assigning a rank.
Our lab signal layer prioritizes independent datasets: specifically Finnrick Analytics, which has tested 6,195 samples from 185 vendors across 15 popular peptides, and Janoshik Analytical, which provides individual task-based verification. The COA layer requires batch-specific artifacts, not static PDFs reused across product pages. If a supplier cannot produce a COA that matches your specific vial's lot number, that is a failure. The trust layer compares discussions from Trustpilot, Reddit (r/Peptides, r/semaglutide), and long-running Telegram communities such as STG and Peptide Research Group. Fulfillment and pricing layers ensure a supplier is not only technically credible but operationally usable for repeat buyers. I also weight whether a vendor accepts credit card payments. Maintaining a merchant account requires passing payment processor compliance checks that Zelle-only or crypto-only vendors have not cleared.
| Criterion | Weight | How scored |
|---|---|---|
| Finnrick Analytics grade | 30% | Independent or community-referenced third-party testing signals with letter-grade context and sample depth. |
| COA transparency | 20% | Batch-specific records, clear lot matching expectations, and verifiable report metadata. |
| Community trust | 20% | Cross-platform reputation patterns from Trustpilot, Reddit, Telegram, and long-running forum threads. |
| Fulfillment reliability | 15% | Shipping consistency, tracking quality, packaging reliability, and support response experience. |
| Pricing transparency | 15% | Value scored against verification quality, not simply lowest list price. |
This methodology is designed for monthly refresh cycles. If new test data, batch failures, or policy regressions surface, ranks can move. That volatility is normal in an unregulated market, which is exactly why static yearly listicles fail users.
One of the most important insights from Finnrick data is that vendor quality is compound-specific: a vendor earning an A rating on BPC-157 may score a D on CJC-1295. I reviewed Finnrick's April 10, 2026 vendor and peptide-level rankings directly, then pulled the strongest current entries for each compound shown here so these tables reflect batch-sensitive leaderboards rather than a single sitewide vendor assumption.
| Rank | Vendor | Finnrick Grade |
|---|---|---|
| 1 | Peptide Partners | A (GREAT) |
| 2 | Orbitrex Peptides | A (GREAT) |
| 3 | Noble Dragons | A (GREAT) |
| 4 | Lipeptides | A (GREAT) |
| Rank | Vendor | Finnrick Grade |
|---|---|---|
| 1 | Orbitrex Peptides | A (GREAT) |
| 2 | Polaris Peptides | A (GREAT) |
| 3 | Aavant Research | A (GREAT) |
| 4 | Shanghai JinBei Chemical | A (GREAT) |
| 5 | Zhengzhou Lanyun | A (GREAT) |
| Rank | Vendor | Finnrick Grade |
|---|---|---|
| 1 | Peptide Crafters | A (GREAT) |
| 2 | Astro Peptides | A (GREAT) |
| 3 | Skye Peptides | A (GREAT) |
| 4 | Amino Amigos | A (GREAT) |
| 5 | Planet Peptide | A (GREAT) |
| Rank | Vendor | Finnrick Grade |
|---|---|---|
| 1 | Peptide Partners | A (GREAT) |
| 2 | Peptide Sciences (defunct) | A (GREAT) |
| 3 | Limitless Life Nootropics | B (GOOD) |
| Rank | Vendor | Finnrick Grade |
|---|---|---|
| 1 | Peptide Partners | A (GREAT) |
| 2 | Peptide Sciences (defunct) | A (GREAT) |
| 3 | Yiwu Aozuo Trading Co | A (GREAT) |
Data source: Finnrick Analytics, accessed April 10, 2026. Ratings are based on tested samples and can change as new batches are submitted. Always verify current ratings before purchasing.[9]
This data demonstrates why blanket vendor recommendations are misleading. A buyer sourcing retatrutide and a buyer sourcing CJC-1295 face completely different vendor quality landscapes. Use these per-compound tables alongside the overall rankings above and the Paradigm Peptides review when you need deeper context on the current retatrutide leader set.
For dosing protocols and reconstitution math, review the tirzepatide protocol, the semaglutide protocol, the BPC-157 protocol, and the ipamorelin protocol. For broader planning, use the PDP protocol library, review PepPal's TB-500 cancer risk analysis when comparing adjacent repair compounds, and use the PepPal reconstitution calculator before finalizing your dilution math.
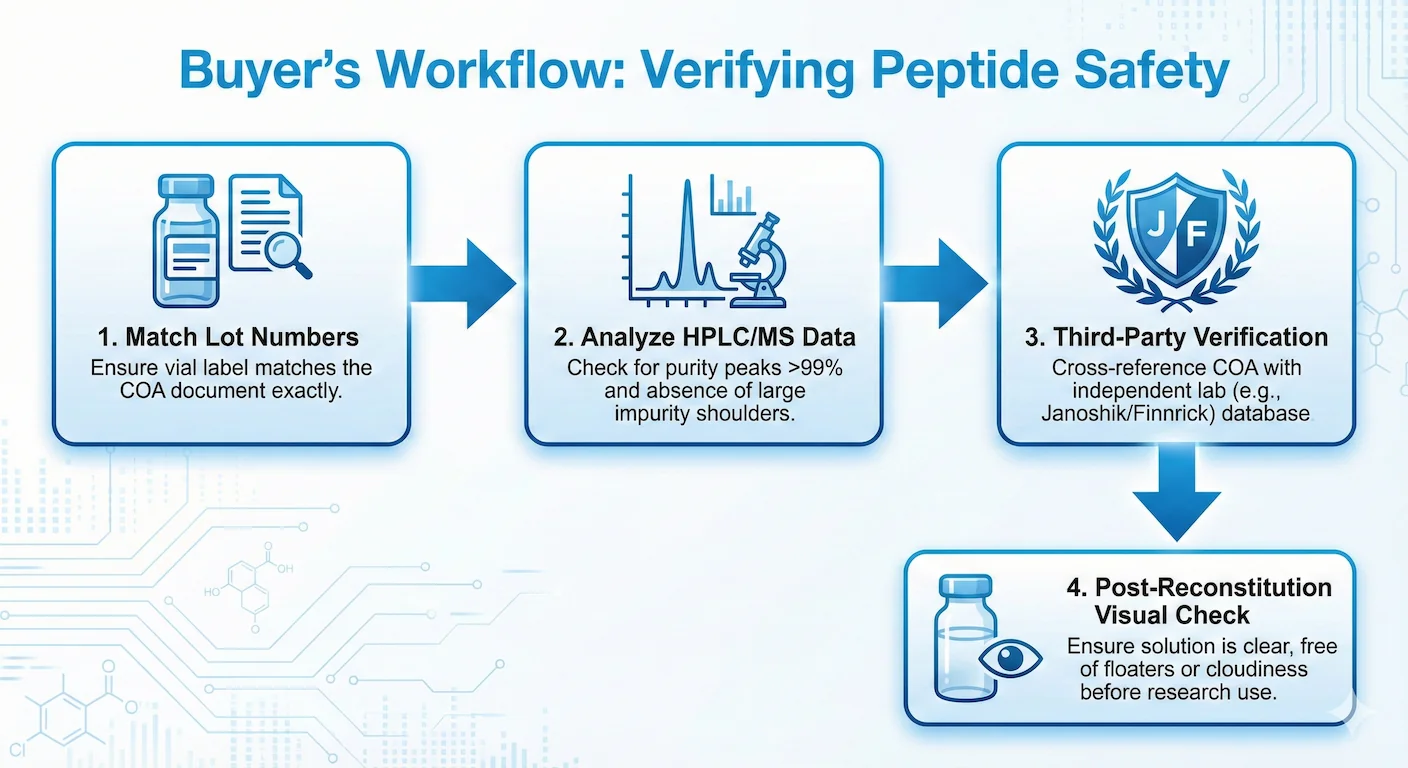
A COA is only useful if you can verify that it belongs to your batch, reflects relevant methods, and matches an independent record trail. Start by checking whether the lot number on your vial matches the lot number on the report. If the supplier sends one generic PDF for every product, treat that as a major trust failure.
When I review supplier documentation for PepPal, I look for lot-matched COAs, method coverage, third-party lab identity, and whether the same evidence can be cross-checked against Finnrick, Janoshik, or a consistent batch-history pattern before I treat the vendor as credible.
Next, confirm method coverage. HPLC can estimate purity, but mass spectrometry is critical for identity confirmation. High purity percentages can still hide harmful impurity profiles if residual solvents, endotoxin contamination, or degradation by products are not assessed. This is why buyers should avoid reducing quality checks to one headline number.
Third-party verification adds needed distance from supplier marketing. Common references in this niche include Finnrick Analytics snapshots and Janoshik task verification workflows. Finnrick Analytics has tested 6,195 samples from 185 vendors across 15 popular peptides as of 2026, making it the largest independent peptide testing dataset publicly available.[4]Their data revealed significant quality variance across the market. For example, Peptide Sciences' retatrutide received a failing E rating across 37 tested samples, while their BPC-157 scored an A, demonstrating that vendor quality is product-specific, not uniform.[5] Community vetting channels such as STG and Peptide Research Group can also surface pattern-level issues faster than isolated reviews because they aggregate repeat buyer experiences and cross-batch observations.
Practical inspection still matters after paperwork checks. A clean lyophilized puck, expected dissolution behavior, and clear reconstituted solution with no visible particulates are baseline observations. Any deviation is a stop signal, not a maybe. Remember the sacrificial-vial principle: one passing test or one clean vial does not guarantee every vial in an unregulated stream. Keep verification as a recurring protocol, not a one-off task.
Once quality checks pass, calculate concentrations and units before handling solvent. Use PepPal’s calculator and follow the full reconstitution protocol to avoid concentration and unit errors.
Community discussion on r/Peptides, r/semaglutide, and long-running Telegram groups like STG and Peptide Research Group consistently surfaces the same themes about grey market sourcing in 2026. To separate repeat signals from one-off anecdotes, I reviewed discussion patterns across threads running from January through April 2026 and weighted recurring verification, fulfillment, and vendor-switching themes more heavily than isolated praise or complaints.[10]
Peptide Partners dominates recommendation threads. Across Reddit supplier comparison posts from January through April 2026, Peptide Partners appears as the most frequently recommended vendor, typically cited for Finnrick sample depth, Janoshik verification, and credit card acceptance. The consistency of these mentions across independent threads and platforms is itself a trust signal.
Pivot Labs is the active PepPal secondary recommendation. Users who recommend alternatives to Peptide Partners most often ask for a practical second option with domestic shipping, usable catalog coverage, and a simple checkout flow. Pivot Labs is the supplier this guide now uses for that role.
The Peptide Sciences closure reshaped community guidance. Post-March 2026 discussion shows a clear migration pattern: experienced users who previously recommended Peptide Sciences shifted primarily toward Peptide Partners and practical secondary suppliers such as Pivot Labs, while newer users increasingly ask about international marketplace options.
Community testing groups are growing. Telegram-based verification groups where members pool independent testing results expanded significantly in 2026. These groups provide real-time batch-level quality context that supplements Finnrick and Janoshik reports.
Common community advice aligns with this guide's methodology: verify every batch independently, never assume one clean order guarantees the next, credit card acceptance matters, and extreme pricing in either direction is a flag.
For more on community verification practices, see how to read a peptide COA.
Safety-aware content matters because this is a YMYL-adjacent topic. The most common contamination concern is endotoxin burden from uncontrolled manufacturing or handling environments. Endotoxins are not always visible and may not be captured by limited reporting formats, so buyers should prefer suppliers with explicit testing standards and replace-on-fail policies.
Heavy metal exposure and residual solvents are additional risk vectors when sourcing standards are inconsistent. Synthesis pathways can leave trace contaminants if purification controls are weak. Even when purity percentages look strong, unresolved solvent residue or non-target compounds can undermine actual quality. This is why method transparency and independent verification outrank marketing language.
Degradation risk is also practical, not theoretical. Peptides can degrade when storage, transport temperature, light exposure, or handling conditions drift from stable ranges. Cold chain failures and repeated temperature cycling increase uncertainty. Fulfillment reliability therefore contributes directly to safety, not just convenience.
Dosing errors are a separate but equally serious category. Community incident reports repeatedly show unit confusion, especially when buyers mix milligrams, micrograms, and syringe units without validated concentration math. The most effective prevention is procedural: calculate first, label clearly, and double-check every conversion before handling solution.
Another overlooked risk is aggregation. Some impurity profiles, including dimeric contamination patterns, may not be obvious in simplified reports and can be missed if buyers over-rely on one metric. This is why a complete verification stack should include identity confirmation, impurity context, and supplier-level consistency over time rather than one successful order.
For additional background on regulatory safety framing, review FDA resources on compounded or unapproved products at FDA.gov and scientific quality-control literature through ScienceDirect.
1. Generic COAs. If the same document appears across unrelated products or batches, documentation is likely decorative rather than traceable.
2. Chat-app-only support. Telegram or WhatsApp-only workflows with no stable support channel make accountability difficult when orders fail.
3. Stock laboratory images. Reverse-search suspicious visuals. Reused stock graphics across multiple vendors are a common trust failure pattern.
4. No refund or replacement policy. High-trust suppliers publish remediation terms for failed lots or fulfillment issues.
5. Very new domain with no identity continuity. Domain age alone is not decisive, but frequent domain changes and missing business identity data are high-risk signals.
6. Therapeutic claims on RUO products. If a supplier markets disease-treatment outcomes directly, compliance discipline is likely weak.
7. Prices far below expected synthesis economics. Extreme discounting often correlates with quality shortcuts or unreliable inventory behavior.
8. Unverifiable business address. Cross-check public business records and mapped location data to avoid shell identities.
9. Frequent domain changes. A vendor that rotates domains repeatedly without clear migration history can be harder to track, verify, and hold accountable.

This comparison drives major search volume because buyers are weighing cost, access, and oversight at the same time. Compounding pharmacies operate under regulated pathways, prescription controls, and facility standards that are not mirrored by grey market suppliers. Grey market channels often reduce friction and price, but they transfer verification duties directly to the buyer.
| Dimension | Grey market | Compounding pharmacy |
|---|---|---|
| Regulatory oversight | Limited; buyer-driven verification | Structured (503A/503B and inspection frameworks) |
| Prescription requirement | Generally no | Yes |
| Typical pricing | Lower upfront, higher verification burden | Higher upfront, stronger process controls |
| Best fit | Advanced buyers willing to verify every batch | Buyers prioritizing regulated oversight |
Neither path is set-and-forget. In both channels, quality and documentation should be treated as active checks. For grey market sourcing, that means using this comparison guide plus your own validation workflow every order cycle.

Get notified when new sourcing explainers, COA breakdowns, and calculator-linked research guides go live. Same lightweight signup flow as PDP, with page-source tracking on each submission.
Email signup
One field. PepPal blog updates, supplier research, and calculator workflows only.
Grey market peptide suppliers usually sell products as research-use-only materials, which places them in a regulatory gray zone. That does not make products approved for human use. Buyers still assume significant quality and compliance risk and must verify documentation independently.
Start with a batch-specific COA tied to your vial lot. Confirm test methods include HPLC and mass spectrometry, then verify the report ID directly with the issuing lab when possible. Purity percentage alone is not enough without identity confirmation and impurity context.
Finnrick Analytics is a third-party peptide testing platform that publishes quality snapshots and letter-grade style assessments based on sampled products. Their data can help compare vendors, but grades can change as new batches are tested, so always re-check current results.
No practical difference. "Grey" and "gray" are spelling variants for the same concept: products sold outside standard regulated pharmaceutical channels but not necessarily through purely illegal counterfeit networks. Verification and safety concerns remain the same either way.
Research use only means the product is marketed for laboratory or analytical use, not for diagnosis, treatment, or personal medical use. Ethical suppliers avoid therapeutic claims and focus on documentation, handling conditions, and traceability for research buyers.
After verifying supplier documentation and vial labeling, calculate concentration targets before adding solvent. Use sterile technique, correct units, and storage controls. PepPal provides a calculator and a full reconstitution guide to reduce common unit and dilution errors.
Yes. Buyers can submit samples to independent analytical labs or cross-check vendor documentation against trusted third-party datasets. Independent testing is one of the strongest safeguards in an unregulated market, but a single passing sample does not guarantee every future batch.
Compounding pharmacies operate under regulated frameworks such as 503A or 503B and require prescriptions for patient-specific use. Grey market suppliers do not follow the same oversight model and generally sell for research use only, usually at lower cost but higher verification burden.
The biggest risks are contamination, mislabeling, inconsistent potency, weak traceability, and dosing mistakes caused by poor instructions or unit confusion. These risks increase when suppliers provide generic COAs, unverifiable test claims, or limited customer accountability.
Pricing varies by peptide complexity, synthesis scale, and testing rigor. Extremely low pricing often signals quality shortcuts, while higher pricing does not guarantee quality on its own. The best value usually comes from suppliers with transparent batch data and reliable fulfillment history.
Peptide Sciences voluntarily shut down on March 6, 2026 without advance warning. The company had been one of the largest grey market research peptide vendors in the United States, reportedly generating approximately $7.4 million in monthly sales. No official explanation was provided beyond the word "voluntary." The closure coincided with escalating FDA enforcement actions, ITC exclusion orders on imported peptides, and Finnrick testing that flagged quality issues with several Peptide Sciences products. Finnrick Analytics has warned that any site still selling under the Peptide Sciences name after March 6, 2026 is fraudulent. Researchers formerly sourcing from Peptide Sciences should verify quality documentation from any new vendor using the supplier comparison table above and the PepPal calculator to confirm reconstitution math before use.
Most reputable grey market peptide suppliers accept credit cards, and credit card acceptance is itself a meaningful trust signal. Maintaining a merchant account requires passing payment processor compliance checks. Processors conduct due diligence on the business, which adds an accountability layer that crypto-only or Zelle-only vendors do not have. All five top-ranked suppliers in this guide accept major credit cards. When a supplier only accepts cryptocurrency, wire transfer, or peer-to-peer payment apps, buyers lose chargeback protection and the vendor faces less accountability for fulfillment failures. This does not mean crypto-only vendors are always bad, but it does mean the buyer carries more risk.
The grey market contracted significantly in 2026. Peptide Sciences, previously the largest vendor, shut down in March. Amino Asylum was raided by the FDA in 2025. At least seven vendors closed in the past twelve months. At the same time, the February 2026 HHS announcement restored approximately 14 peptides to Category 1 compounding status, creating a regulated access channel through licensed compounding pharmacies with a prescription. The combination of shrinking grey market supply and expanding regulated access means buyers now have more options but also face a more complex decision framework. For a deeper look at the regulatory developments, see the FDA enforcement coverage and the RFK Jr. peptide reclassification analysis on PepPal News.
PepPal's supplier quality data comes primarily from Finnrick Analytics, an independent third-party peptide testing platform that has tested over 6,195 samples from 185 vendors. Regulatory claims reference FDA.gov directly, including the agency's published safety page on unapproved GLP-1 drugs and the FDA warning letters database. Legal context is sourced from published analyses by law firms including Wilson Sonsini and Frier Levitt. Community reputation data draws from Reddit (r/Peptides), Trustpilot, and established Telegram groups. The full source hierarchy and research methodology is described on the PepPal About page. Every claim in this guide is traceable to the sources listed below the FAQ.
Based on Finnrick Analytics data as of May 2026, the top-rated vendors vary by compound. For retatrutide, Paradigm Peptide, Orbitrex Peptides, and Peptide Partners all hold A (GREAT) ratings. For tirzepatide, Polaris Peptides and Aavant Research lead. For BPC-157, Peptide Partners holds the top position. Finnrick has tested over 6,195 samples from 185 vendors, making it the largest independent peptide testing dataset available. However, grades are compound-specific and batch-dependent, so always check current Finnrick data for your specific peptide before ordering. See the per-compound Finnrick tables above for the full breakdown.
After Peptide Sciences voluntarily shut down on March 6, 2026, the strongest alternatives depend on what you valued. For testing depth, Peptide Partners now has 59 Finnrick-tested samples across 7 products, exceeding Peptide Sciences' verified sample count. For catalog breadth and practical replacement coverage, Pivot Labs is now the active PepPal secondary pick because it combines a usable research peptide catalog with domestic shipping, PEPPAL code support, and a straightforward checkout workflow. Biotech Peptides remains relevant for maximum category coverage. For budget-conscious buyers, Paradigm Peptides and Core Peptides offer lower per-vial pricing. Finnrick Analytics flagged Peptide Sciences' retatrutide with a failing E rating across 37 samples before the closure, so the former gold-standard reputation was already eroding. See the full ranked comparison above.
Across r/Peptides and r/semaglutide discussion threads from January through April 2026, Peptide Partners is the most consistently recommended grey market vendor, typically cited for Finnrick sample depth, Janoshik-verified COAs, and credit card payment acceptance. Pivot Labs is PepPal's current secondary recommendation for practical catalog coverage, domestic shipping, and straightforward ordering. Community consensus also emphasizes that no vendor recommendation replaces batch-level verification, so experienced Reddit users still advise checking Finnrick data for your specific compound and requesting lot-matched COAs before purchasing.
International marketplace platforms like Peptaura connect buyers with Chinese manufacturers at significantly lower prices, often 50-70% below US-based vendors. However, the savings come with substantial additional risk: longer shipping times, uncertain cold-chain management during transit, limited recourse for failed orders, and higher contamination risk without verified clean-room standards. Community testing groups show variable quality across marketplace sellers. PepPal does not recommend this route for most buyers. If cost is your primary concern, domestic vendors like Paradigm Peptides and Core Peptides offer competitive pricing with shorter shipping, credit card protection, and easier COA verification. Experienced researchers can use international sourcing, but only with strict batch-level verification on every order.
Preferred supplier
Peptide PartnersNeed peptides? Start with a verified supplier.
PepPal's recommended source with current discount access and established testing standards.
The following sources support the regulatory, quality, and market claims made in this guide. PepPal follows a published source hierarchy described on the About page . Regulatory filings and independent third-party data are prioritized over supplier marketing and community discussion.
[1] Wilson Sonsini Goodrich & Rosati, "FDA Sends Warning Letters to More Than 50 GLP-1 Compounders and Manufacturers," October 2025.. 1] Wilson Sonsini Goodrich & Rosati, "FDA Sends Warning Letters to More Than 50 GLP-1 Compounders and Manufacturers," October 2025. https://www.wsgr.com/en/insights/fda-sends-warning-letters-to-more-than-50-glp-1-compounders-and-manufacturers.html
[2] U.S. Food and Drug Administration, "FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss," updated February 2026. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fdas-concerns-unapproved-glp-1-drugs-used-weight-loss
[3] U.S. Food and Drug Administration, Warning Letters to research peptide vendors, 2024–2026. Referenced in Frier Levitt, "Regulatory Status of Peptide Compounding in 2025," January 2026. https://www.frierlevitt.com/articles/regulatory-status-of-peptide-compounding-in-2025/
[4] Finnrick Analytics, independent peptide testing platform. 6,195 samples tested from 185 vendors across 15 peptides. https://www.finnrick.com/
[5] Finnrick Analytics, Peptide Sciences vendor testing data, December 2024–March 2026. Retatrutide: E rating (37 samples). BPC-157: A rating. Referenced in multiple industry analyses including BioStrata Research and Peptide Publicus. https://www.finnrick.com/vendors/peptide-sciences
[6] United States v. Matthew Kawa (Paradigm Peptides), U.S. Attorney's Office, Northern District of Indiana, December 2025. Guilty pleas entered December 10, 2025. Referenced in industry reporting by The Peptide Catalog and Peptide Publicus. https://www.justice.gov/usao-ndin/case/us-v-matthew-kawa-et-al
[7] U.S. Food and Drug Administration, "FDA clarifies policies for compounders as national GLP-1 supply begins to stabilize," updated 2025. https://www.fda.gov/drugs/drug-safety-and-availability/fda-clarifies-policies-compounders-national-glp-1-supply-begins-stabilize
[8] Frier Levitt LLC, "Regulatory Status of Peptide Compounding in 2025," January 2026. Legal analysis of 503A/503B compounding frameworks and FDA enforcement posture. https://www.frierlevitt.com/articles/regulatory-status-of-peptide-compounding-in-2025/
[9] Finnrick Analytics, Per-Compound Vendor Rankings, April 2026. Tirzepatide, Semaglutide, Retatrutide, BPC-157, CJC-1295, and Ipamorelin leaderboards. https://www.finnrick.com/
[10] Reddit r/Peptides community, supplier recommendation threads, January-April 2026. Aggregated community discussion patterns. https://www.reddit.com/r/Peptides/
If you are actively comparing vendors, start with the supplier directory for profile-level details on each ranked vendor, then validate batch documentation and planned concentrations before ordering.
Before making a final choice, learn how to read a peptide COA so you can grade documentation quality with the same framework PepPal uses. If you are sourcing for a specific goal, the use-case guides can help narrow your compound selection: fat loss peptides, muscle and recovery peptides, or skin and collagen peptides.
If you are price-comparing after narrowing the field, review PepPal's discount codes before checkout so you are comparing final out-of-pocket cost rather than list price alone.
Once you have selected a supplier and received your vials, use the free PepPal reconstitution calculator to get exact syringe units for any vial size and BAC water volume, then follow the step-by-step reconstitution guide to prepare safely. If you are planning to combine multiple compounds, review Peptide Stacking 101 for safety considerations.